|
|
Post by 1dave on Feb 5, 2023 1:16:49 GMT -5
I know that one's not...but my wife is constantly showing me pictures on Facebook of "amazing" clouds...and I think the vast majority of them are Photoshopped. It used to be pictures were "proof" of something...but those days are over. I LOVE seeing shapes in clouds...or rocks...or Rorschach Tests! LOL Why do psychiatrists have so many pictures of BATS? |
|
|
|
Post by 1dave on Feb 5, 2023 1:03:14 GMT -5
Do you enjoy finding interesting cloud shapes?  |
|
|
|
Post by 1dave on Feb 5, 2023 0:31:10 GMT -5
According to all the studies, Yellowstone is past due eruption time. Can we do anything about it? There is a study on the differences between the volcanoes leading to Yellowstone, and those to the south in the great Basin. link.springer.com/article/10.1007/s00445-007-0138-1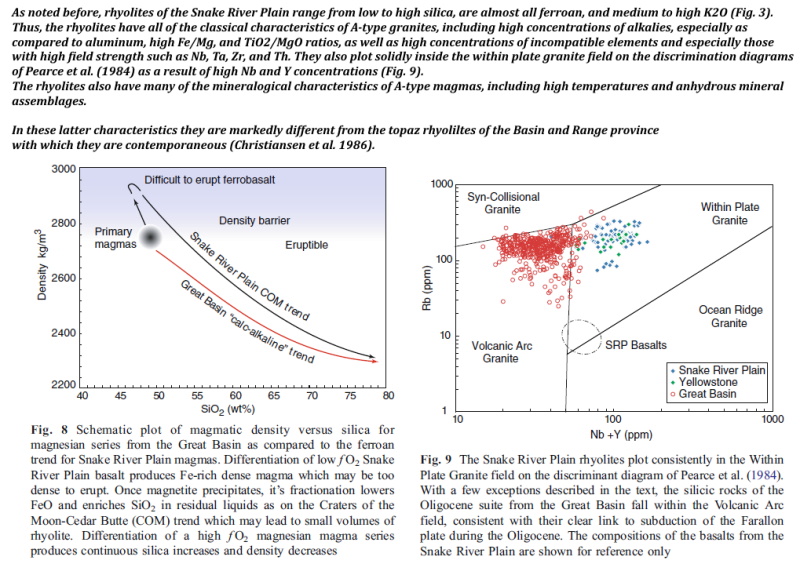 Note top left " 3000 Difficult to erupt ferobasalt." Magmas normally evolve from heavy high iron basalt to light silica rhyolite icon_think What if Environmentalists were to begin dumping scrap iron into all of the Yellowstone geysers, converting the highly eruptible rhyolite back to difficult heavy basalt?  |
|
|
|
Post by 1dave on Feb 3, 2023 20:55:51 GMT -5
Gander Why is Gander slang for look? Definition of take a gander at Look at, glance at, as in Will you take a gander at that woman's red hair! This slangy idiom, dating from the early 1900s, presumably came from the verb gander, meaning “stretch one's neck to see,” possibly alluding to the long neck of the male goose. For a synonym, see take a look at. bramblewoodhill.com/gender-goose-geese/  |
|
|
|
Post by 1dave on Feb 3, 2023 11:59:07 GMT -5
 Thanks Beth and Jason for helping me find the good threads! "Simple SiO2" is AMA-ZING-LY complicated! Especially with water. 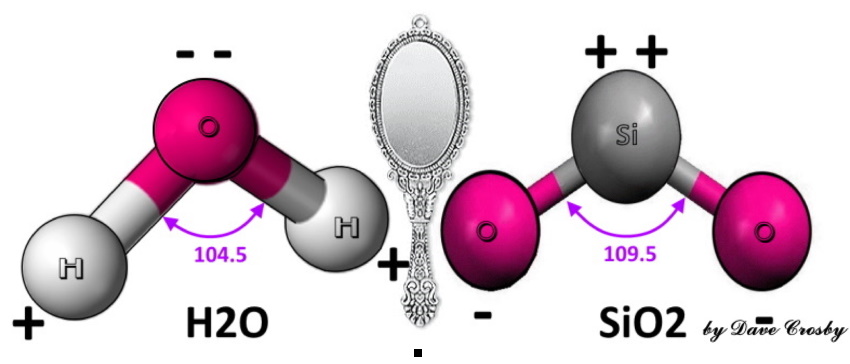 Water is held together by weak hydrogen or pi bonds, easily breaks apart - "ionizes," while Quartz is held together by strong multiple lattice covalent bonds.  Very Low (acid) and Very High (base) pH will eat the flesh off your bones.
Acid will dissolve calcite and similar elements, but base will force them to be deposited.
On the other hand, Base will dissolve a little silica, while acid forces silica to be deposited.
Even in very high pH, SiO2 is not very soluble. Why low solubility? Because silicon atoms are larger than carbon atoms, silicon-oxygen bonds are longer than C02 carbon-oxygen equivalents. The p orbitals on silicon and oxygen cannot overlap enough to form weak pi bonds. Therefore, only single strong covalent bonds are formed. So each silicon atom forms four single covalent bonds with four oxygen atoms, and each oxygen atom forms two single covalent bonds with two silicon atoms. With each of the silicon atoms bridged to its four neighbors via an oxygen atom also attached to the neighbor silicon atoms, this forms a large network covalent structure. Strong bonds in three dimensions make silicon dioxide a hard, high melting point solid with a structure similar to that of diamonds. The bond energy is estimated at 621.7 kJ/mol. To top it off, MOST SUBSTANCES ARE HYDROPHILIC (water loving), while SiO2 in most forms is HYDROPHOBIC! That is, they have H- or OH- negative surfaces that repel negative H2O. Where do those occasional huge amounts of silica in solution come from? That answer came from JOHN ELLISTON in Australia! Shock waves shatter rocks - AND SILICA BONDS! Ground water ionizes, grabs those newly freed Si bonds, become rivers of Si-H or Si-OH fibers and start filling in voids. Agate is made. There you have it. Most Agate layers were formed back when earth had it’s worst shocks, the dying times! Agate is periodically a sedimentary rock ! NOTE: Normally in different high pH locations Alkaline and Alkaline Earth elements are deposited and replaced in the groundwater with only minor amounts of silica. Much of New Agate is held together by weak hydrogen bonds! As the agate dries the water evaporates and the hydrogen bonds are replaced as Si and O make new strong connections! This is why the heat treatment hardens flint for napping. 
|
|
|
|
Post by 1dave on Feb 3, 2023 2:01:44 GMT -5
|
|
|
|
Post by 1dave on Feb 2, 2023 12:41:45 GMT -5
THE BOOK on John Day Fm. etc. Thundereggs! "Geological Survey Bulletin 1161-D 1964" I have searched for this for YEARS and could not find it. I didn't know he made it into a book! This morning I found THIS on Amazon! www.amazon.com/gp/product/1289107831/ref=ppx_yo_dt_b_asin_image_o00_s00?ie=UTF8&psc=1 Temporarily out of stock but I ordered it! Happy as a June bug in a corn field! 
I strongly disagree with his theory on T.E. Formation -  |
|
|
|
Post by 1dave on Feb 1, 2023 19:24:19 GMT -5
|
|
|
|
Post by 1dave on Feb 1, 2023 11:43:53 GMT -5
Welcome from Northern Arizona! You'll really like hanging out here.  I have to agree with Robin. The Best New-bees are the busy sharing Bees.  |
|
|
|
Post by 1dave on Feb 1, 2023 11:23:34 GMT -5
|
|
|
|
Post by 1dave on Feb 1, 2023 9:37:39 GMT -5
|
|
|
|
Post by 1dave on Feb 1, 2023 8:50:38 GMT -5
Thanks jamesp, I'm so glad to have you as a friend to bounce ideas of of! I LIKE that perspective. This morning I'm thinking of a row of silicon motels on a busy highway with H- and OH+ water molecules racing by. Each motel has 4 vacancies.  If it fills up with OH+ customers, the owner happily watches the cash flow in and is homophobically satisfied. If it is on an acidic street, only H- customers drive by and it is homophilic, in and out customers. Does that work? |
|
|
|
Post by 1dave on Feb 1, 2023 2:13:22 GMT -5
Hello three ! You have only been here for three hours, so I haven't been able to discern whether you are very young or old for your age. Perhaps you can explain? In the meantime,  |
|
|
|
Post by 1dave on Feb 1, 2023 1:54:48 GMT -5
|
|
|
|
Post by 1dave on Feb 1, 2023 0:03:52 GMT -5
jamesp I'm trying to understand a new concept: Hydrophobic Rocks!   the pH of the hydrolyzing compound dictates the degree to which it is hydrophilic or hydrophobic. Acidic pH produces hydrophilic siloxanes. In a more alkaline environment, the siloxane will tend to be more hydrophobic. Once the Si-O bonding type is formed (either hydrophobic or hydrophilic), later acidification or alkaline change does not alter the nature of the Si-O bonding. The dissolved SiO exhibits a significant vapor pressure, and when cooled will nucleate to form sub-micronic fibroids which remain hydrophobic. with hydrogen Si-OH. As simple monomers and dimers, these compounds are normally hydrophilic and referred to as siloxanes or silanols (the silicon analogs to ketones and alcohols). In polymeric form, these cross-linked silicon-oxygen bonded compounds are generally hydrophobic and referred to as silicones.  It needs a lot of new drawings to make heads or tails of. icon_think
|
|
|
|
Post by 1dave on Jan 31, 2023 12:56:23 GMT -5
|
|
|
|
Post by 1dave on Jan 31, 2023 10:58:44 GMT -5
The SiO2 bonds are POWERFUL, forcing other minerals to the center to crystalize last.
|
|
|
|
Post by 1dave on Jan 31, 2023 7:34:53 GMT -5
OH! Surprise!  Well, the photo is really all I need.  |
|
|
|
Post by 1dave on Jan 31, 2023 7:01:28 GMT -5
I went shopping for rhyolite cabs this morning -  AND bought this tiny Thunderegg!  |
|
|
|
Post by 1dave on Jan 30, 2023 23:13:11 GMT -5
Too bad it is photoed pages instead of a regular PDF, but the images are fantastic. My only disagreement is they are convinced that thundereggs developed in ash fall tuffs as feldspars. That is not possible. Thundereggs indisputably formed as multi layered cristobalite needles separated by feldspar coatings crystal spheres. NOT feldspars. That requires lots of movement, time and high temperatures not available after an ash flow tuff is in place.  IF the needles were to get in place, how would the water get into the mashed flat layer to explode the needles apart? No, it just doesn't work. I have found thundereggs in ash flow tuffs, but they had to form in the magma chamber long before eruption! The bond energy of 621.7 kJ/mol explains how the spheres stayed together during flight. Remember they were in the middle of a high pressure explosive cloud. any near the edge with low pressure would explode like hand grenades and all trace of them lost in the chaos. Those that survived were mashed flat along with steam vessels etc.that surrounded them.   |
|