nursetumbler
Cave Dweller 
Member since February 2022
Posts: 928 
|
Post by nursetumbler on Sept 5, 2022 10:14:49 GMT -5
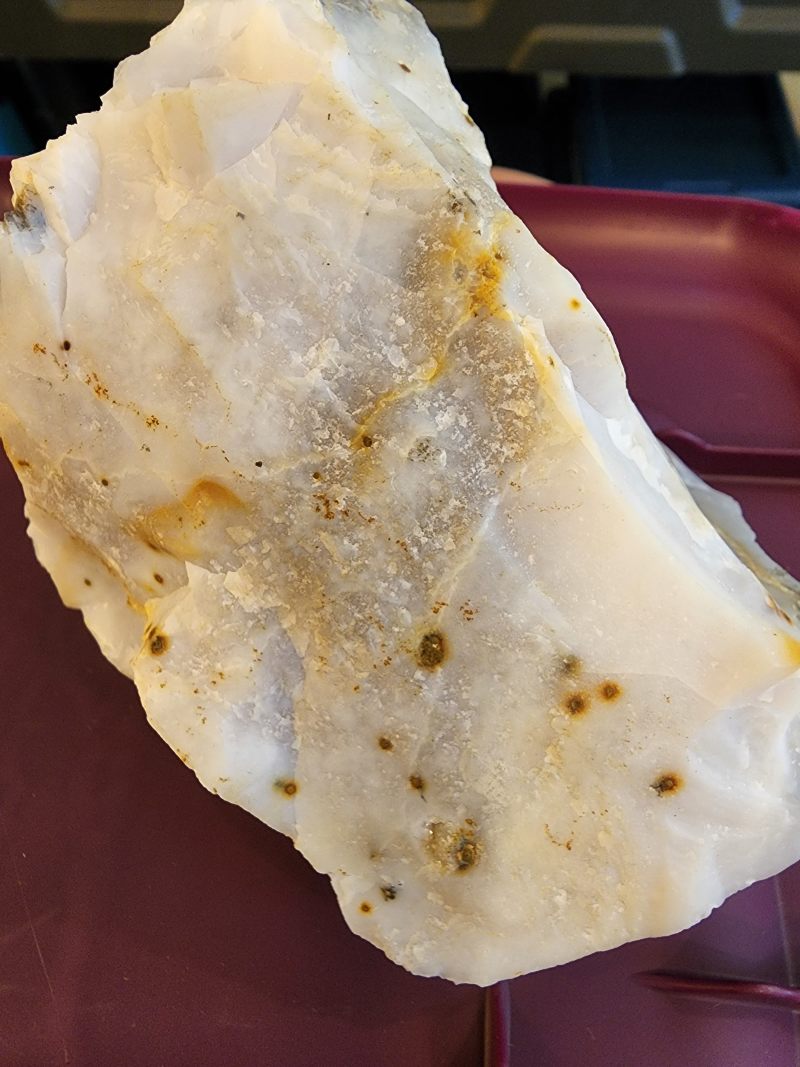  The spots seem to be iron inclusions. |
|
Tommy
Administrator 
Member since January 2013
Posts: 12,652
|
Post by Tommy on Sept 5, 2022 10:40:55 GMT -5
Looks more like quartz or quartzite. Chalcedony should be basically clear like agate but without internal inclusions.
|
|
nursetumbler
Cave Dweller 
Member since February 2022
Posts: 928 
|
Post by nursetumbler on Sept 5, 2022 16:04:50 GMT -5
Looks more like quartz or quartzite. Chalcedony should be basically clear like agate but without internal inclusions. Thank you Tommy |
|
|
|
Post by vegasjames on Sept 5, 2022 16:34:33 GMT -5
Quartz. You can see the crystal structure. Chalcedonies are cryptocrystalline to microcrystalline and thus you cannot see the crystal structure by the naked eye.
I discuss this in this post from another board:
I responded to a post here earlier with various misinformation such as claiming agate is amorphous like opal and that opal is a chalcedony. Since agates are a chalcedony and all chalcedonies are crystalline, then agate cannot be amorphous like opal since amorphous means without a crystal structure.:
"Opal is not a chalcedony. Opal converts in to a chalcedony. Chalcedonies are microcrystalline or cryptocrystalline quartz. Both cryptocrystalline and microcrystalline are in essence the same thing in the fact that the crystals are so small that they cannot be seen with the naked eye. The only difference is that under microscope magnification the crystals of microcrystalline quartz can be differentiated. Cryptocrystalline quartz crystals on the other hand are so small that under microscope magnification the crystals cannot be differentiated. There are actually around 300 types of opal, and not al are stones. The stingers of bees and of nettle plants are covered with opal. And bamboo sap, which is super high in silica, can form a type of opal called "vegetable opal". Opals we refer to stones though start out from saturated to supersaturated silica solutions. I won't bother going in to a ton of details, but the silica molecules are attracted towards each other through forces known as van der Waals forces. As these molecules move closer together, water is squeezed out forming a solid amorphous (no crystalline structure) gel. When the water content reaches as average of 3-21% the result is opal, which again is not a true solid, but rather a solid gel. As opal continues to lose water, below 3%, the opal converts from an amorphous gel, to some form of chalcedony (common chalcedony, agate, jasper or chert/flint) all of which are microcrystalline or cryptocrystalline quartz (silicon dioxide), none of which are amorphous since they all have crystal structure unlike opal. What form of chalcedony the opal with take will depend on the silica source. Agates and common jaspers are formed from inorganic silica sources such as sandstone or clays. Chert/flint, which is actually a subcategory of jasper is produced from opal created from silica derived from the biogenic sources of diatoms and radiolarians. Opal varies quite a bit in hardness and stability for a couple of reasons. One is water content. The higher the water content of opal the softer it is, as well as the less stable it is. The stability issue goes back to the fact that opal is not a true solid, but rather a solid gel. Therefore, it does not really have a stable structure. As water is lost from the opal, the gel much restructure to gain some sense of stability. If water is lost from the opal too rapidly, then the opal does not have the time to properly restructure and the opal will form cracks (craze). To stabilize high water content opals, the water content much be very slowly reduced over 6 months to years out of light and with a fairly consistent temperature. The second factor is aluminum oxide content. Most people think opals are just pure silicon dioxide and water, which is very rarely the case. Opals can contain various other elements and compounds such as uranium, barite. The primary other component of most opal though is aluminum oxide. The aluminum oxide content to opal affects not only its density, but also hardness that both increase with increasing aluminum oxide content. The aluminum oxide is mainly derived from clays, but can also come from other sources such as diatoms. As a final note, contrary to what is commonly taught, opal does not take millions of years to form. The formation of opal has been shown to rather rapid, and can be grown and stabilized in as little as a year. Its conversion in to chalcedonies such as agate or jasper does take significantly more time."
|
|
ericabelle
spending too much on rocks
  Instagram acct: @erica_shoots_everything
Instagram acct: @erica_shoots_everything
Member since April 2021
Posts: 482
|
Post by ericabelle on Sept 5, 2022 20:59:29 GMT -5
Looks like quartzite with some iron staining to me
|
|
|
|
Post by Rockoonz on Sept 5, 2022 21:32:33 GMT -5
Looks like polka dot agate. I think more like quartz/quartzite with iron stained vugs rather than the oolite like tiny concretions (?) in polkadot. |
|
|
|
Post by Rockoonz on Sept 6, 2022 12:37:59 GMT -5
Definitely similar but the iron staining and spots that look more like vugs are definitely not found in PDA.
|
|
|
|
Post by Rockoonz on Sept 6, 2022 17:45:54 GMT -5
Bravura Cabochon DesignsYour example of PDA, yes absolutely. The agate nursetumbler asked about 100% definitely not PDA. Iron may be some small part of what creates the color on the polka-dots, but it is never iron so solidly ferrous that it would leave the iron staining on the surface.
|
|